Know Your Patients
Test Their Systemic Chronic Inflammation
After more than 20 years of scientific research and use across Europe as a leading biomarker for systemic chronic inflammation, the suPARbio™ Systemic Chronic Inflammation Test has arrived in the United States — available soon under CLIA regulations for Laboratory Developed Tests (LDTs).
It has not been cleared or approved by the U.S. Food and Drug Administration. At this time, this test is not available for residents of the State of New York.
suPARbio™ Systemic Chronic Inflammation Test
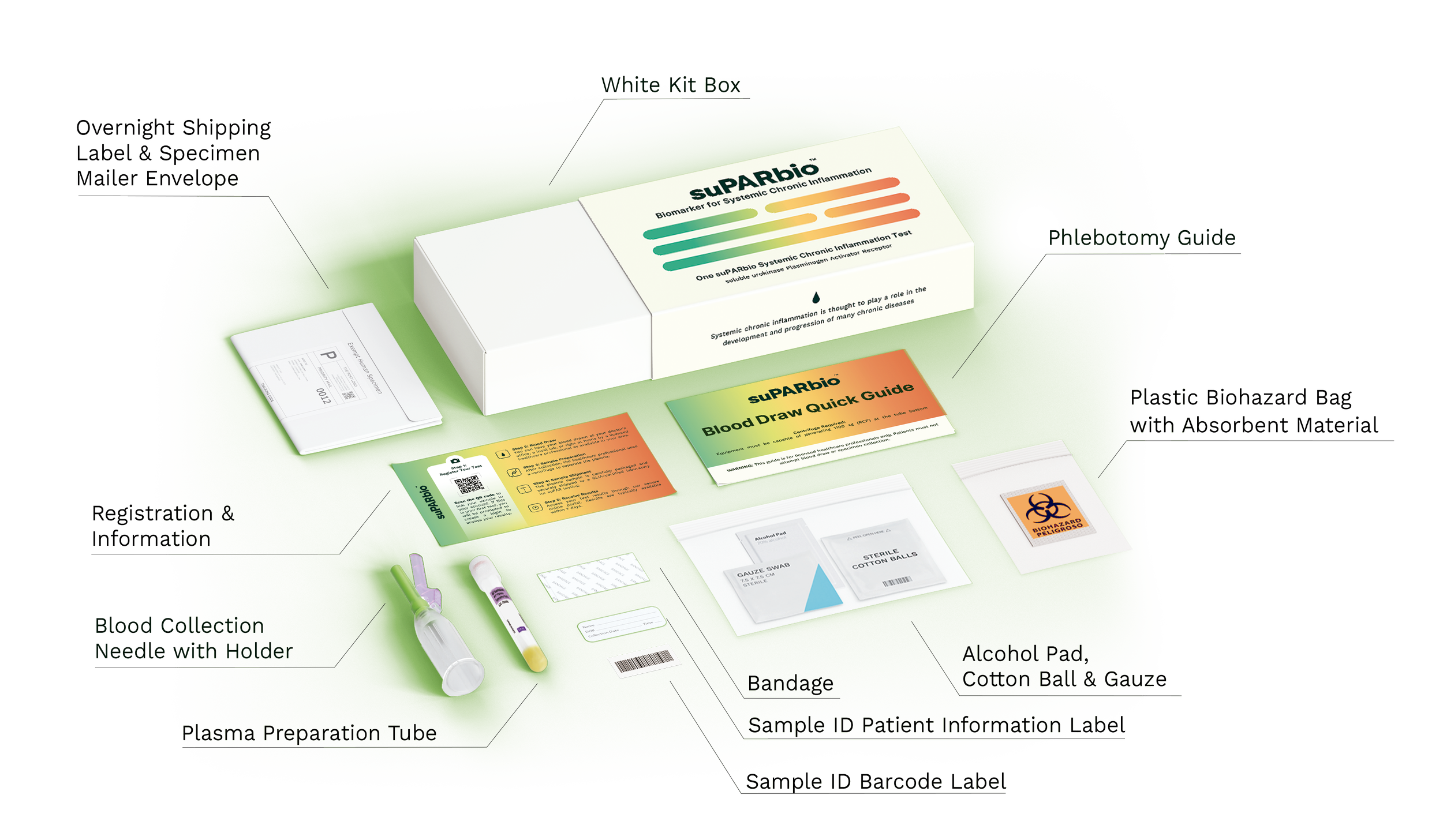
Kit Components
The suPARbio™ Systemic Chronic Inflammation Test kit is carefully assembled and includes everything needed for a safe, professional collection and shipment to our laboratory.
Each kit is designed for:
Professional use — intended for use by qualified healthcare professionals to enable simple and secure plasma collection following a standard blood draw.
Safe, reliable, pre-paid shipping — all materials comply with applicable medical specimen transport standards.
Simple registration — scan the included QR code prior to the blood draw to register your kit and link your sample to your secure account.
Your kit includes:
A step-by-step Blood Draw & Shipping Guide
Pre-labeled sample collection and return materials
suPARbio-branded packaging design to support a clean and professional handling experience
This test has been validated as a Laboratory Developed Test (LDT) under CLIA regulations. It has not been cleared or approved by the U.S. Food and Drug Administration (FDA) and is not intended to diagnose, treat, cure, or prevent any disease or medical condition. Test results should be interpreted by a qualified healthcare provider in the context of the patient’s clinical history, symptoms, and other laboratory findings. At this time, this test is not available for residents of the State of New York.
